
PROSTATE CANCER DIAGNOSTICS

Prostate Cancer is the Silent Killer - There's Still No Screening...
Prostate Cancer is the Silent Killer - There's Still No Screening...
We're Changing That!
We're Changing That!
Prostate Cancer (PCa)
Prostate cancer is the most commonly diagnosed cancer in men in the UK, with over 56,000 new cases recorded in 2020—a figure projected to rise to more than 75,000 by 2040.
The good news is that survival rates are high when the disease is caught early: over 99% of patients survive through the first three stages of prostate cancer. However, survival drops significantly once the disease progresses to stage 4, highlighting the critical importance of early detection and ongoing surveillance.
With both incidence and mortality rates on the rise, demand for effective prostate cancer screening is expected to grow rapidly. In the UK alone, 12,000 men die from prostate cancer every year, and 1 in 8 men will be diagnosed during their lifetime.
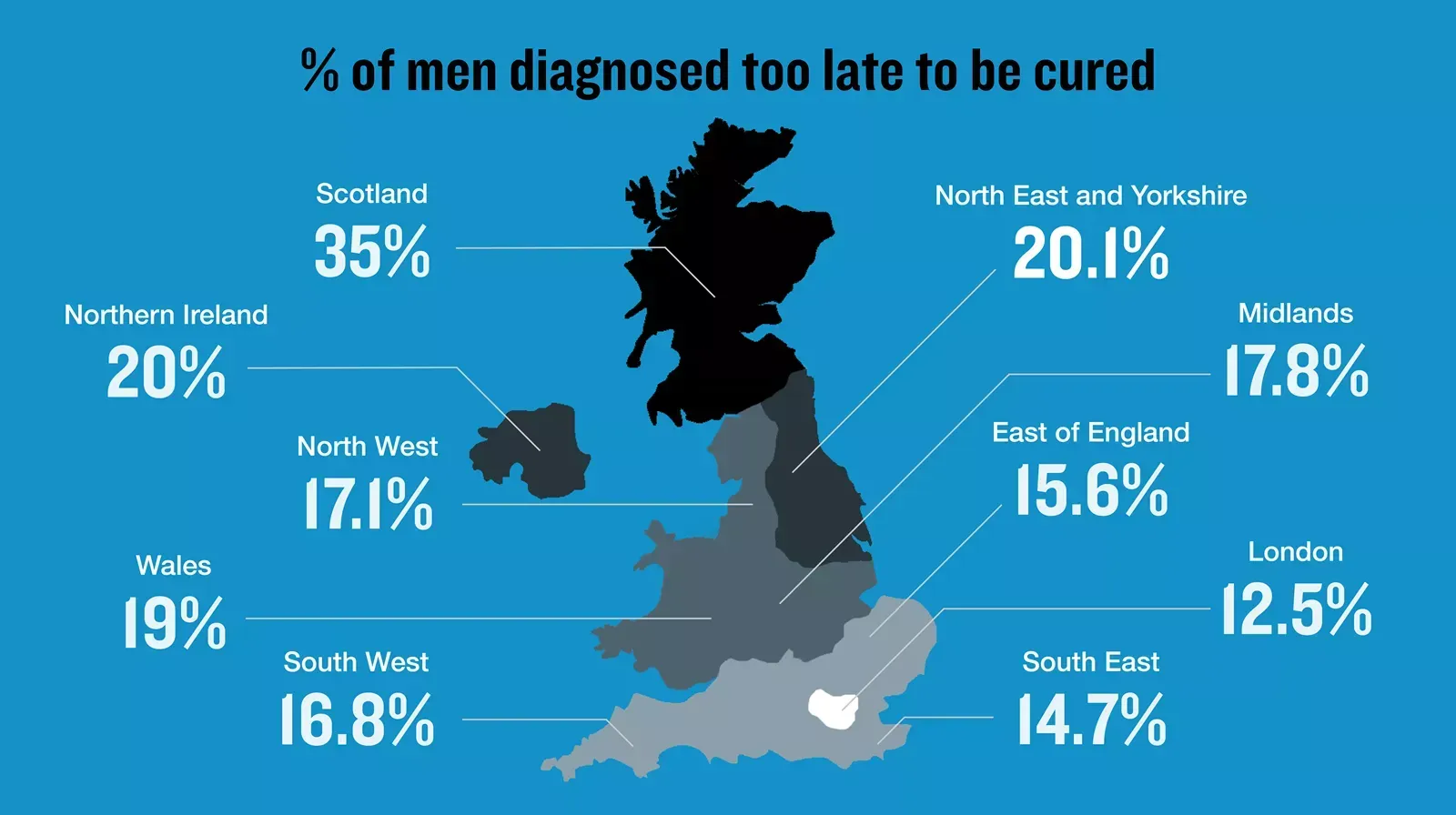
In Scotland, the situation is particularly urgent: the percentage of men diagnosed too late for curative treatment is more than double that of other parts of the UK.
Prostate Cancer (PCa)
Prostate cancer is the most commonly diagnosed cancer in men in the UK, with over 56,000 new cases recorded in 2020—a figure projected to rise to more than 75,000 by 2040.
The good news is that survival rates are high when the disease is caught early: over 99% of patients survive through the first three stages of prostate cancer. However, survival drops significantly once the disease progresses to stage 4, highlighting the critical importance of early detection and ongoing surveillance.
With both incidence and mortality rates on the rise, demand for effective prostate cancer screening is expected to grow rapidly. In the UK alone, 12,000 men die from prostate cancer every year, and 1 in 8 men will be diagnosed during their lifetime.
In Scotland, the situation is particularly urgent: the percentage of men diagnosed too late for curative treatment is more than double that of other parts of the UK.
The Second Most Common
Cancer in Men
Prostate cancer is the second most common cancer in men globally, and the fourth most common cancer overall. In 2022, approximately 1.47 million new cases were recorded worldwide, with the highest incidence rates in countries such as Guadeloupe, Lithuania, and Norway. Each year, an estimated 1.5 million men are diagnosed, and the disease leads to over 397,000 deaths globally.
Delays Are Costing Lives
In the UK, long waiting times for diagnosis and treatment are compounding the crisis. As of April 2024:
- Just 73.5% of people received confirmation of a cancer diagnosis within 28 days of an urgent referral.
- Only 66.6% of patients began treatment within 62 days of an urgent cancer referral.
These delays can have devastating consequences. When it comes to prostate cancer, early diagnosis and timely treatment are critical to survival—and far too many men are being diagnosed too late.
The Second Most Common
Cancer in Men
Prostate cancer is the second most common cancer in men globally, and the fourth most common cancer overall. In 2022, approximately 1.47 million new cases were recorded worldwide, with the highest incidence rates in countries such as Guadeloupe, Lithuania, and Norway. Each year, an estimated 1.5 million men are diagnosed, and the disease leads to over 397,000 deaths globally.

Delays Are Costing Lives
In the UK, long waiting times for diagnosis and treatment are compounding the crisis. As of April 2024:
- Just 73.5% of people received confirmation of a cancer diagnosis within 28 days of an urgent referral.
- Only 66.6% of patients began treatment within 62 days of an urgent cancer referral.
These delays can have devastating consequences. When it comes to prostate cancer, early diagnosis and timely treatment are critical to survival—and far too many men are being diagnosed too late.








Background
The current state-of-the-art screening method for prostate cancer—the prostate-specific antigen (PSA) test—relies on a blood sample to measure PSA levels. However, PSA is a highly variable biomarker, influenced by a range of non-cancer-related factors such as age, infection, and prostate enlargement. As a result, PSA testing suffers from high false-positive rates and low specificity, limiting its overall diagnostic accuracy.
Due to these limitations, clinicians are often forced to rely on more invasive diagnostic procedures, which carry risks of bleeding, infection, and patient discomfort—as well as a significant cost burden to healthcare systems.
Background
The current state-of-the-art screening method for prostate cancer—the prostate-specific antigen (PSA) test—relies on a blood sample to measure PSA levels. However, PSA is a highly variable biomarker, influenced by a range of non-cancer-related factors such as age, infection, and prostate enlargement. As a result, PSA testing suffers from high false-positive rates and low specificity, limiting its overall diagnostic accuracy.
Due to these limitations, clinicians are often forced to rely on more invasive diagnostic procedures, which carry risks of bleeding, infection, and patient discomfort—as well as a significant cost burden to healthcare systems.
Therefore, currently Clinicians are dependent on more intrusive procedures which put the patients at risk of bleeding, wounds and infections, coupled with the large associated cost.
Background
The current state-of-the-art screening method for prostate cancer—the prostate-specific antigen (PSA) test—relies on a blood sample to measure PSA levels. However, PSA is a highly variable biomarker, influenced by a range of non-cancer-related factors such as age, infection, and prostate enlargement. As a result, PSA testing suffers from high false-positive rates and low specificity, limiting its overall diagnostic accuracy.
Due to these limitations, clinicians are often forced to rely on more invasive diagnostic procedures, which carry risks of bleeding, infection, and patient discomfort—as well as a significant cost burden to healthcare systems.
Therefore, currently Clinicians are dependent on more intrusive procedures which put the patients at risk of bleeding, wounds and infections, coupled with the large associated cost.
Developing the Solution
In 2020 alone, approximately 500,000 men underwent potentially unnecessary biopsies due to inaccurate prostate cancer testing. While multiparametric MRI (mpMRI) has emerged as a tool to support clinical decision-making, it is labour-intensive, expensive, and not widely accessible—limiting its effectiveness as a scalable screening method.
At the heart of this issue is a glaring gap: there is currently no prostate cancer (PCa) screening program anywhere in the world.
Existing diagnostic approaches are invasive, uncomfortable, and highly inconvenient—and they routinely fail to detect the disease early, particularly in older men, who are at highest risk. These limitations underscore the urgent need for a reliable, accessible, large-scale screening solution that can transform how prostate cancer is detected and managed.
Pinpoint Medical is developing that solution.
Developing the Solution
In 2020 alone, approximately 500,000 men underwent potentially unnecessary biopsies due to inaccurate prostate cancer testing. While multiparametric MRI (mpMRI) has emerged as a tool to support clinical decision-making, it is labour-intensive, expensive, and not widely accessible—limiting its effectiveness as a scalable screening method.
At the heart of this issue is a glaring gap: there is currently no prostate cancer (PCa) screening program anywhere in the world.
Existing diagnostic approaches are invasive, uncomfortable, and highly inconvenient—and they routinely fail to detect the disease early, particularly in older men, who are at highest risk. These limitations underscore the urgent need for a reliable, accessible, large-scale screening solution that can transform how prostate cancer is detected and managed.
Pinpoint Medical is developing that solution.
Early Detection, Better Outcomes – The Future of Cancer Screening Starts Here.

The Solution
Pinpoint Medical’s PCD is a non-invasive, urine-based screening tool for prostate cancer that delivers high specificity while maintaining strong sensitivity—ensuring accurate detection without the discomfort or complexity of traditional methods.
A Simple, Patient-Friendly Process:
- Urine Sample Collection
The patient provides a standard urine sample, eliminating the need for blood tests or invasive procedures.
- Biomarker Measurement
The system detects specific biomarkers in the urine that indicate the presence of prostate cancer.
- Rapid Results
In just 15 minutes, PCD delivers clear, actionable results—enabling fast, efficient screening at the point of care.
This simplicity and speed make PCD an ideal solution for widespread prostate cancer screening, ensuring more men are tested early and can receive timely, potentially life-saving treatment.
The Solution
Pinpoint Medical’s PCD is a non-invasive, urine-based screening tool for prostate cancer that delivers high specificity while maintaining strong sensitivity—ensuring accurate detection without the discomfort or complexity of traditional methods.
A Simple, Patient-Friendly Process:
- Urine Sample Collection
The patient provides a standard urine sample, eliminating the need for blood tests or invasive procedures.
- Biomarker Measurement
The system detects specific biomarkers in the urine that indicate the presence of prostate cancer.
- Rapid Results
In just 15 minutes, PCD delivers clear, actionable results—enabling fast, efficient screening at the point of care.
This simplicity and speed make PCD an ideal solution for widespread prostate cancer screening, ensuring more men are tested early and can receive timely, potentially life-saving treatment.

The Solution
Pinpoint Medical’s PCD is a non-invasive, urine-based screening tool for prostate cancer that delivers high specificity while maintaining strong sensitivity—ensuring accurate detection without the discomfort or complexity of traditional methods.
A Simple, Patient-Friendly Process:
- Urine Sample Collection
The patient provides a standard urine sample, eliminating the need for blood tests or invasive procedures.
- Biomarker Measurement
The system detects specific biomarkers in the urine that indicate the presence of prostate cancer.
- Rapid Results
In just 15 minutes, PCD delivers clear, actionable results—enabling fast, efficient screening at the point of care.
This simplicity and speed make PCD an ideal solution for widespread prostate cancer screening, ensuring more men are tested early and can receive timely, potentially life-saving treatment.

Progress with PCD has been exceptional, and we are confident that market barriers will rapidly diminish as we continue to onboard key partners and investors.
We’ve already achieved successful clinical validation in Hong Kong SAR, with further validation underway in the UK and additional regions. In the UK, we have secured a 500-patient test case study in collaboration with the Beatson Institute for Cancer Research in Glasgow—a major milestone toward broader adoption.
Our market analysis identifies a global target market of approximately 800 million men, representing a significant opportunity for impact and scalability.
Accessible. Scalable. System-Changing.
PCD’s non-invasive test kit eliminates the need for trained clinicians or laboratory settings. This enables routine screening of high-risk individuals in local clinics and general practices—easing the burden on healthcare systems while improving early detection and survivability rates.
Smart Tech Meets Public Health
PCD is supported by a cloud-based digital platform that delivers instant test results to both patients and healthcare providers. Beyond clinical utility, the platform offers robust capabilities for large-scale data collection and analysis—enabling insights into population-level risk, geographic trends, and informing evidence-based government policy.
Progress with PCD has been exceptional, and we are confident that market barriers will rapidly diminish as we continue to onboard key partners and investors.
We’ve already achieved successful clinical validation in Hong Kong SAR, with further validation underway in the UK and additional regions. In the UK, we have secured a 500-patient test case study in collaboration with the Beatson Institute for Cancer Research in Glasgow—a major milestone toward broader adoption.
Our market analysis identifies a global target market of approximately 800 million men, representing a significant opportunity for impact and scalability.
Accessible. Scalable. System-Changing.
PCD’s non-invasive test kit eliminates the need for trained clinicians or laboratory settings. This enables routine screening of high-risk individuals in local clinics and general practices—easing the burden on healthcare systems while improving early detection and survivability rates.
Smart Tech Meets Public Health
PCD is supported by a cloud-based digital platform that delivers instant test results to both patients and healthcare providers. Beyond clinical utility, the platform offers robust capabilities for large-scale data collection and analysis—enabling insights into population-level risk, geographic trends, and informing evidence-based government policy.
Generation II

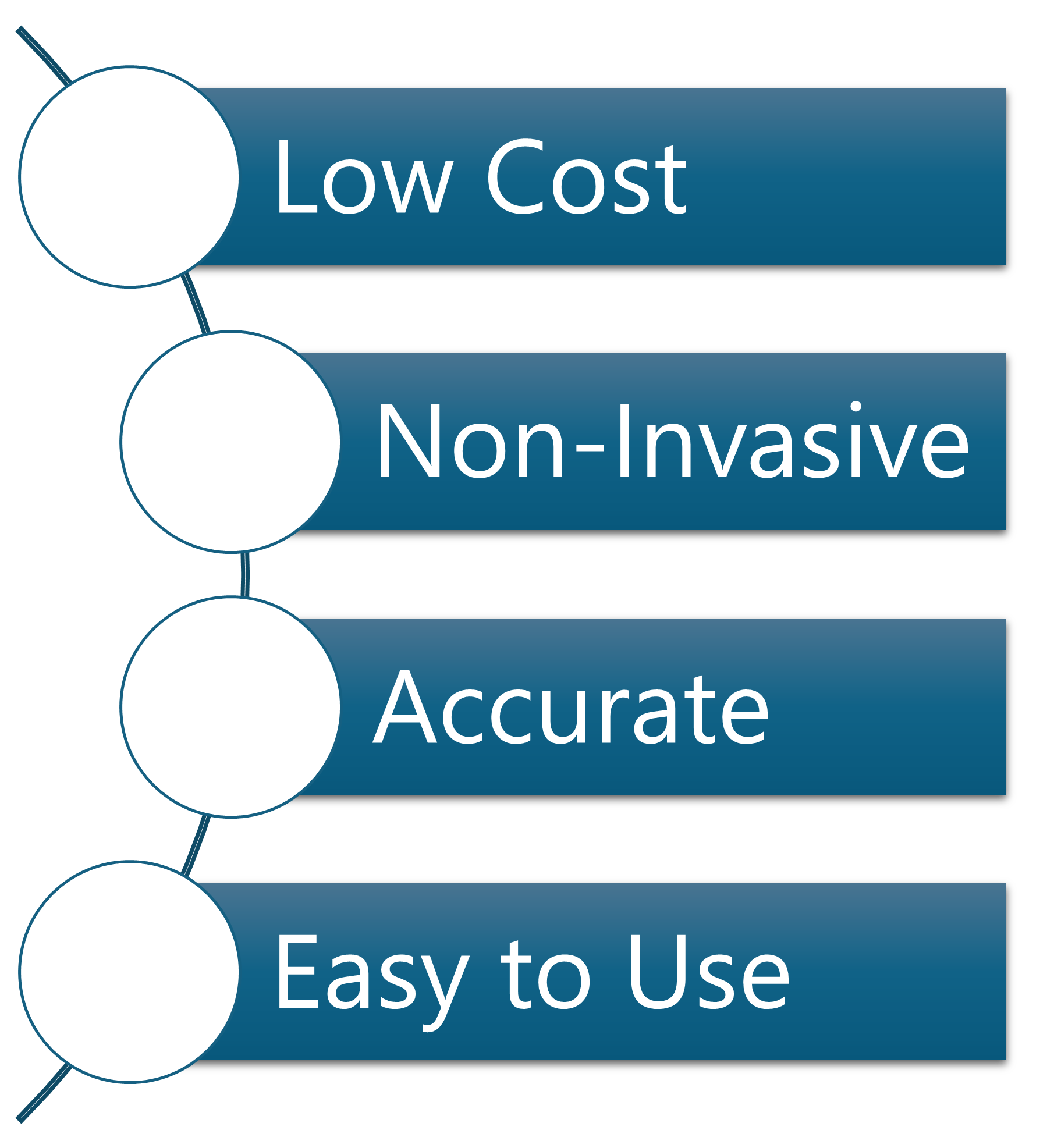
- Cost-Effective – Affordable to scale across primary care settings.
- Non-Invasive – Urine-based testing eliminates discomfort and risk.
- Highly Accurate – Maintains strong specificity and sensitivity.
- Simple & User-Friendly- Designed for ease of use by patients and clinicians alike.

- Cost-Effective – Affordable to scale across primary care settings.
- Non-Invasive – Urine-based testing eliminates discomfort and risk.
- Highly Accurate – Maintains strong specificity and sensitivity.
- Simple & User-Friendly- Designed for ease of use by patients and clinicians alike.

Professor Roy Vellaisamy
Gold Medal Winner with his “Sensor Platform”
International Exhibition of Inventions Geneva 2019
Professor Roy Vellaisamy
Gold Medal Winner with his
“Sensor Platform”
International Exhibition of Inventions Geneva 2019
Point of Care Devices-from Food Safety to Non-Invasive Early Cancer Detection
“Point of Care Devi ces-from Food Safety to Non-Invasive Early Cancer Detection” developed by Professor Roy Vellaisamy, Associate Professor in the Department of Materials Science and Engineering (MSE) at City University of Hong Kong, who is also the member of State Key of Laboratory of Terahertz and Millimeter Waves, and research team. Approximately 1% of the global population suffers from histamine intolerance. Histamine can be found when bacteria grows in food, so the existence of histamine is one of the indicators of food decay. However, conventional tests for histamine take about one day in a laboratory and the majority of people do not have access to
affordable onsite testing devices.

“Point of Care Devi ces-from Food Safety to Non-Invasive Early Cancer Detection” developed by Professor Roy Vellaisamy, Associate Professor in the Department of Materials Science and Engineering (MSE) at City University of Hong Kong, who is also the member of State Key of Laboratory of Terahertz and Millimeter Waves, and research team. Approximately 1% of the global population suffers from histamine intolerance. Histamine can be found when bacteria grows in food, so the existence of histamine is one of the indicators of food decay. However, conventional tests for histamine take about one day in a laboratory and the majority of people do not have access to affordable onsite testing devices.


Recognising Excellence: Celebrating Innovation in Biosensing Technology.
Originally Designed for the
Detection of Contaminants in Food
The new system developed by the CityU research team was originally designed for thedetection of contaminants in food, with concentrations of less than 0.2ppm (parts per million). It contains a nano-sensor that can prescreen food samples on the spot using asmartphone apps to identify the presence of histamine and formaldehyde in 10 to 25 minutes.
The research team goes a step further by modifying the point-of-care sensing platform to detect cancer biomarkers, such as conducting non-invasive urine tests to screen for prostate cancer biomarkers. This approach helps increase the survival rate of PCa patients as early detection is crucial to increase the chance of successful treatment.
Originally Designed for the Detection of Contaminants in Food


Commited to Innovation
Dr. Affar S. Karimullah - Chief Technology Officer
“While significant progress has been made in breast cancer screening, it’s deeply concerning that we’ve yet to achieve the same for one of the most common cancers affecting men. Our award-winning technology offers a long-overdue solution for prostate cancer screening and diagnostics. At Pinpoint Medical, we’re confident that we can change the landscape—enabling earlier detection, improving survival rates, reducing healthcare costs, and ultimately saving lives.”
Dr. Affar S. Karimullah
Chief Technology Officer, Pinpoint Medical

PCD is a groundbreaking, non-invasive prostate cancer screening solution—clinically validated, scalable, and ready to disrupt a global market of over 800 million men.
We’re seeking strategic investment partners to help bring this life-saving technology to market. With rapid testing, unmatched accessibility, and real-world validation already underway, PCD is positioned for global adoption and exponential growth.
If you're looking to invest in innovation with real clinical impact and commercial potential, we’d love to hear from you.
Jason Donnelly, Managing Director, PinPoint Medical Ltd.

PCD is a groundbreaking, non-invasive prostate cancer screening solution—clinically validated, scalable, and ready to disrupt a global market of over 800 million men.
We’re seeking strategic investment partners to help bring this life-saving technology to market. With rapid testing, unmatched accessibility, and real-world validation already underway, PCD is positioned for global adoption and exponential growth.
If you're looking to invest in innovation with real clinical impact and commercial potential, we’d love to hear from you.
Jason Donnelly, Managing Director, PinPoint Medical Ltd.

CONTACT INFORMATION
Phone: +44 (0)7796 777 739
Email: info@pinpointmedical.co.uk
Address: St Colmes, Ballinluig, Pitlochry, United Kingdom, PH9 0NR
OUR LOCATION
CONTACT INFORMATION
Phone: +44 (0)7796 777 739
Email: info@pinpointmedical.co.uk
